 Home > Nature of light > State of light
Home > Nature of light > State of light
State of light
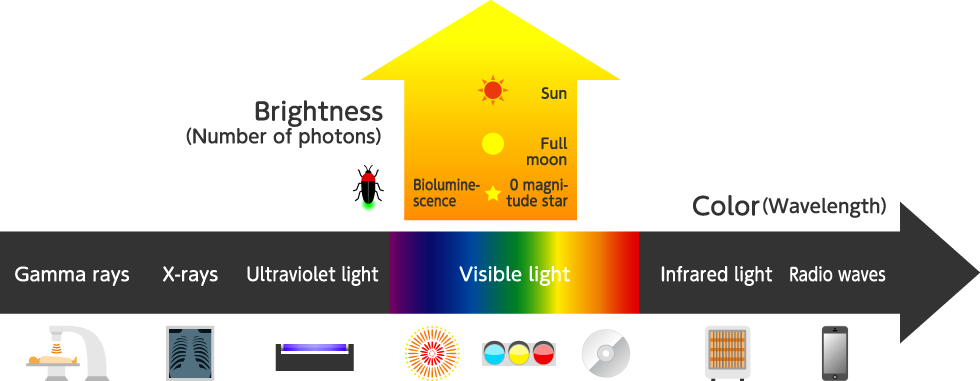
The “wavelength” of light is related to light color and non-visible light, and “the number of photons” is related to the brightness of light. Moreover, light is not only emitted from the sun but also from the bodies of living organisms or lighting equipment.
Let’s take a closer look at the various figures of light.
Color and wavelength
When light is viewed as wave (electromagnetic waves), the color is defined by the wavelength which is the length between crests or between troughs of the wave. The light that can be detected by human eyes is called visible light and is on a wavelength between 400 to 700 nanometers. One nanometer (nm) is equal to one billionth of a meter and is used along with the micrometer (1 μm or 1 millionth of a meter) to express unit of small length.
The electromagnetic wave region including radio waves, terahertz waves and infrared light exists on longer wavelengths than visible light. On the other hand, ultraviolet light, as well as X-rays and gamma rays, exist on shorter wavelengths than visible light.
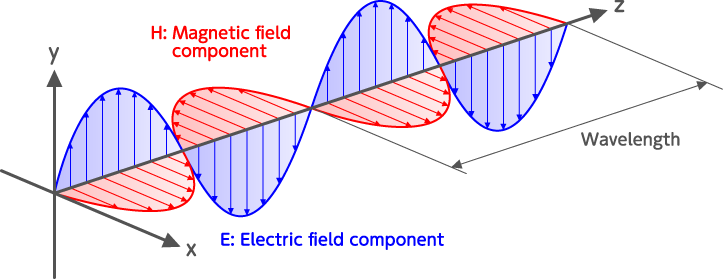
Electromagnetic waves are waves from electric and magnetic fields that oscillate perpendicular to each other and to the direction of propagation. While light has the dual properties of a particle and a wave, electromagnetic wave focuses on the properties of “wave”. So, in quantum mechanics, electromagnetic waves are photons themselves. The distance that an electromagnetic wave propagates during one cycle of the electric or magnetic field oscillation is called the wavelength. Light (electromagnetic wave) travels in a vacuum at a speed of about 300,000 kilometers per second. Therefore, electromagnetic waves whose oscillation cycle is one hundred-trillionth of a second are infrared lights of wavelength 3 μm.
Visible light
Seven colors of the rainbow are visible light
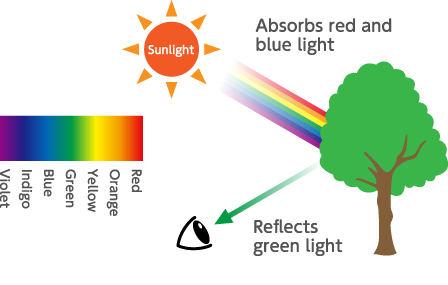
Visible light is light in a wavelength range between about 400 to 700 nanometers. Human eyes can detect the light in this wavelength range. Visible light expressed as color ranges from purple in the vicinity of 400 nm to longer wavelengths, to deep blue, blue, green, yellow and orange and red in the vicinity of 700 nm. Human eyes are most sensitive to green-colored light on the 555 nm wavelength.
The reason that the leaves of plants appear green in sunlight is that substances contained in the leaves soak up blue and red light and reflect back only the green light that reaches our eyes.
Infrared light
Infrared lights are present wherever there is heat
Infrared light presents on a relatively wide wavelength range between 700 nm to 1 mm and are generally categorized as follows.
- Near-infrared ray:700 nm to 2.5 μm
- Mid-infrared ray:2.5 μm to 4 μm
- Far-infrared ray:4 μm to 1000 μm (1 mm)
Near-infrared lights close to visible light are found in devices used in daily life such as remote controllers for home appliances and audio-visual equipment and communications between portable terminals.
Infrared lights are emitted from a heat source (or anything that has heat). For example, the human body at a temperature of about 37 °C constantly emits far-infrared lights around 10 μm. Ear thermometers and thermography can measure human body temperature by detecting the far-infrared emissions.
Ultraviolet light
Birds and insects can see ultraviolet lights
Ultraviolet lights on a short wavelength away from visible light are invisible to humans yet can be seen by birds and insects. As shown in the picture on the right, even flower petals that appear to the human eye to have no patterns, can be seen as having intense patterns in the center section to insects that can see ultraviolet light. Insects can in this way find and swoop down on the nectar from flowers.


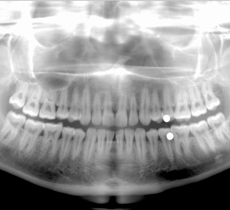
X-rays
Rays that penetrates through various objects
When accelerated electrons drastically decelerate after collision with a material or change their direction of motion in a magnetic field, X-rays, electromagnetic waves of a wavelength between 0.01 to 10 nm, are emitted. X-rays are also generated when the inner orbit of the atom becomes vacant and an electron from an outer orbit makes a transition there.
X-rays on a wavelength of about 0.01 nm can strongly penetrate through objects so are often used in such fields as medicine to capture interior images of the body and manufacturing industry to ensure product quality.

Gamma rays
High energy rays that acts on material
Gamma rays are high-energy electromagnetic waves emitted from the nucleus of atoms in an excitation state. The energy of gamma ray photons is ten thousand to 1 million times greater than the energy possessed by photons in visible light. When gamma rays are irradiated into material, high-speed electrons are generated and acts on the surrounding materials. Gamma ray is used for sterilization and cancer radiation treatments.
Plants getting plenty of sunlight grow quick and healthy. Light is indispensable to growth of plant. However, just as we humans have preference for food, plants also have “preference of light”. Their favorite are blue, red, and near-infrared lights, and are highly sensitive to wavelengths of these types of light.
Plants use light not only for energy source for photosynthesis. They recognize the color of the sky to assess their environment. Plants make use of the differences of light to detect the length from daybreak to sunset in a day (diurnal rhythm) and also to find seasonal changes through amount of sunlight, and set the timing for morphogenesis (period for flowering, coloring of leaves and bearing fruit). For example, it is known that the flavor of farm products is related to the sunset. The flavorful component will faintly change due to the plant’s sensitivity to light. Plants in this way offer us the seasons of the year by their appearance, fragrance, and taste.

Brightness of light
When the light wavelength changes in this way, our eyes sense the difference in “color” to see things as blue or as red. Well then, in the case of bright light or faint light, what kind of difference do our eyes sense? The “photon,” the smallest form of light, is the key to answering that question.
We use units such as illuminance or power density (W/m2) to express brightness of light. Here, let’s try to express brightness of light based on the number of photons. The photon is a particle of light and the energy it contains is determined by the wavelength so the photons gradually appear when made darker.
On a sunny day, the brightness of sunlight in terms of visible light power density is approximately 400 W/m2. Expressing this as the number of photons gives about 1,000,000,000,000,000 photons per second per square millimeter, or in other words about a quadrillion photons per square millimeter, reach to the earth every second. The brightness of a full moon is about 1 millionth that of the sun, and the brightness of a star of zero magnitude is 1 hundred-thousandth that of a full moon. The light emitted from living organisms such as fireflies and bioluminescent jellyfish is about the same level as the brightness of the moon or stars.

Polarization of Light
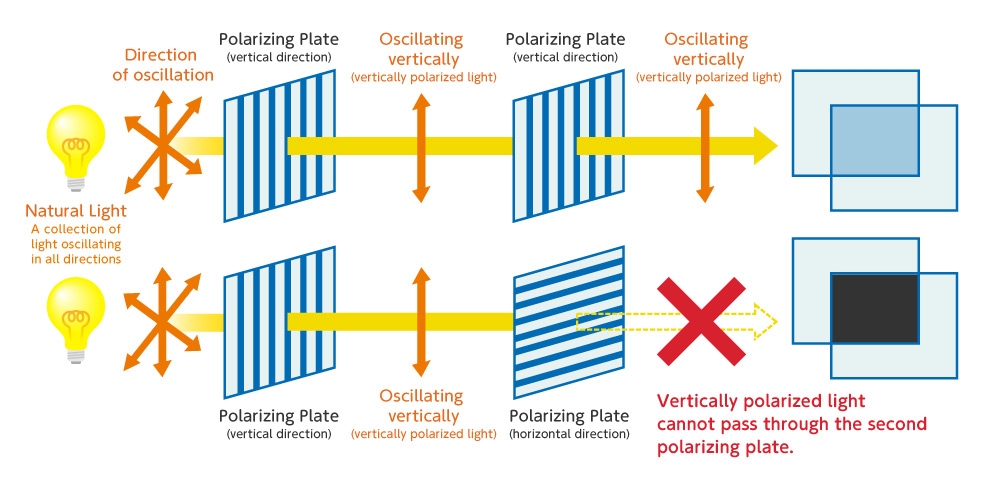
Light is a transverse wave that oscillates in directions perpendicular to the direction it travels. Natural light oscillate 360° in all directions (in the figure, the orange arrow indicates the direction of oscillation). In contrast to this, light which oscillates in only one direction is called polarized light. We make great use of polarized light for all manner of applications.
How is polarized light used? A special plate called a polarizing plate is used to get light that oscillates in only one direction from natural light. When a polarizing plate is placed vertically as shown in the figure below, only light oscillating in the vertical direction can pass through the polarizing plate (vertically polarized light).
When a second polarizing plate is set in the horizontal direction, the vertically polarized light cannot pass through the second polarizing plate, so no light passes through it. (For this reason, it turns pitch black where two polarizing plates are overlapped.)
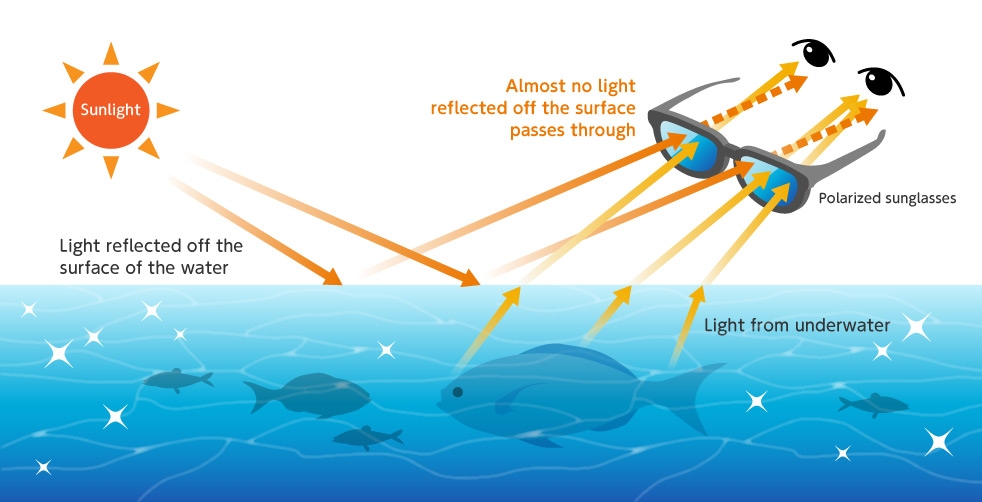
For example, polarized sunglasses for fishing employ this mechanism. The polarized sunglasses cut light reflected off the surface of the water (most of which oscillates parallel to the surface of the water), thereby reducing obstruction of light coming from underwater (reflected glare from the sun), so it is possible to better see fish in the water.
Also, the LCD monitors of TVs, smartphones, and other devices adjust the amount of red, green, and blue light with a polarizing filter in order to create different colors.

Polarized sunglasses
Cuts light reflected from the surface of the water, making it easier to see fish in the water.
LCD monitors for TVs and smartphones
Adjusts the amount of red, green, and blue light with a polarizing filter in order to create different colors.
3D glasses
Viewing pictures with different polarized light on the left and right makes photographs and pictures appear to be 3D.Light from living organisms - bioluminescence -
Light comes not only from the sun. In nature, there are types of lights generated in living organisms such as the lights emitted from firefly and bioluminescent jellyfish. Living organisms emit light for the reasons such as;
- To collect other organisms for feeding
- To scare away enemies
- To communicate with fellow organisms
The light emitted from organism is created by a chemical reaction in their body. A firefly has a luminescent substance called luciferin in its body that reacts with oxygen, causes a transition to a high energy state (energetic state), and the surplus energy is exchanged to the luminescence that is emitted from the body.

Photo courtesy of Kamo Aquarium


Mr. OHSUKA

Mr. TAKAMOTO






