 Home > Photon lab. > Topics > The story of fluorine (F) in connection with IYPT 2019
Home > Photon lab. > Topics > The story of fluorine (F) in connection with IYPT 2019
The story of fluorine (F) in connection with IYPT 2019
Finding out about illness with light!?
The story of fluorine (F) in connection with the International Year of the Periodic Table of Chemical Elements 2019

- Written by
-
Yoshiyuki Otake Central Research Laboratory, Hamamatsu Photonics K.K.
Specialized field : Quantum electronics, quantum optics, optical physics
INDEX
2019 is the International Year of the Periodic Table of Chemical Elements
Finding a small cancer with fluorine and light
Putting “markers” on what cancer cells like
Why does the FDG test drug emit light? What is FDG?
2019 is the International Year of the Periodic Table of Chemical Elements

The year 2019 marks the 150th anniversary of the discovery of the periodic law of the elements by a Russian chemist named Dmitri Mendeleev in 1869. The UNESCO and UN therefore established the International Year of the Periodic Table of Chemical Elements (IYPT 2019), so various types of events linked to this discovery are being held throughout the world at this time.
Even in Japan, through the campaign (website) called Our Elements,we can see essay contests held for junior high school, high school, and college students who will be the scientists of the future and also messages from universities and corporations.
This article takes up the topic of fluorine (symbol F) as an element from the periodic table of chemical elements in connection with the International Year of the Periodic Table of Chemical Elements and also gives a description of how it’s used in medical diagnosis.
Even in Japan, through the campaign (website) called Our Elements,we can see essay contests held for junior high school, high school, and college students who will be the scientists of the future and also messages from universities and corporations.
This article takes up the topic of fluorine (symbol F) as an element from the periodic table of chemical elements in connection with the International Year of the Periodic Table of Chemical Elements and also gives a description of how it’s used in medical diagnosis.
Periodic table of elements
Finding a small cancer with fluorine and light

Cancer is the number one cause of death among all diseases for both men and women in Japan. However, medical treatment is making progress and cancer is a disease that can be cured. The most important thing about cancer treatment is finding it while it is still small. An effective diagnostic technique for finding a small cancer is called PET (positron emission tomography) that makes use of the characteristics of fluorine and light.
The so-called “PET cancer screening” takes advantage of the properties of cancer cells to find a small cancer that was very difficult to detect up to now. The cells of the human body utilize the glucose produced from food and elsewhere as an energy source for daily activities. Cancer cells, however, are characterized by taking in large amounts of glucose compared to normal cells. This means if we can find points with high concentrations of glucose then we can detect cancer.
The so-called “PET cancer screening” takes advantage of the properties of cancer cells to find a small cancer that was very difficult to detect up to now. The cells of the human body utilize the glucose produced from food and elsewhere as an energy source for daily activities. Cancer cells, however, are characterized by taking in large amounts of glucose compared to normal cells. This means if we can find points with high concentrations of glucose then we can detect cancer.
Putting “markers” on what cancer cells like

But, how do we find glucose in a living human body and even glucose concentrated in the tiny cells in the body?
PET diagnosis does this by way of a test drug called FDG. This FDG has nearly the same properties as glucose except that it contains atoms that emit light called gamma rays. So this FDG is a drug with a “marker of light” that is put on the favorite energy source of the cancer cells.
When this FDG is injected into a human body, the cancer cells ingest a large amount of FDG in the same way as they take in glucose, and emit gamma rays. By detecting and measuring these gamma rays with high-sensitivity light sensors placed outside the body, it is possible to search out concentrations of FDG, in other words, the locations where cancer is probably present.
PET diagnosis does this by way of a test drug called FDG. This FDG has nearly the same properties as glucose except that it contains atoms that emit light called gamma rays. So this FDG is a drug with a “marker of light” that is put on the favorite energy source of the cancer cells.
When this FDG is injected into a human body, the cancer cells ingest a large amount of FDG in the same way as they take in glucose, and emit gamma rays. By detecting and measuring these gamma rays with high-sensitivity light sensors placed outside the body, it is possible to search out concentrations of FDG, in other words, the locations where cancer is probably present.
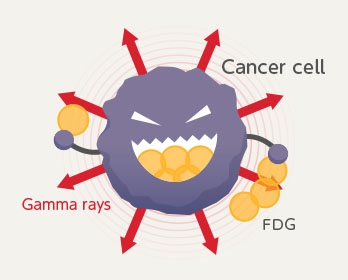
The cancer cell location can be pinpointed by measuring the gamma rays emitted from the FDG taken into the cancer cells.
PET cancer screening
Why does the FDG test drug emit light? What is FDG?

So FDG in this way lets us know where cancer cells are present by emitting gamma rays that serve as a marker of light. But why does FDG emit gamma rays? To find the answer, we have to know what FDG is made from.
The correct name for FDG is 18F-FDG. The FDG is an abbreviation for fluorodeoxyglucose which is its full name in English. The F stands for fluoro (has fluorine), D stands for deoxy (takes in oxygen), and G stands for glucose. The name of the chemical in this way indicates its structure and the name also gives a hint that FDG resembles glucose.
So what is 18F anyway? This 18F apparently holds the secret behind how it works as a “marker of light.” Actually, this 18F is what is called an “isotope” of the fluorine and this isotope is heavily involved with the phenomenon of gamma ray emission. Now let’s dive a little deeper into this topic, though it will be a little more specialized.
The correct name for FDG is 18F-FDG. The FDG is an abbreviation for fluorodeoxyglucose which is its full name in English. The F stands for fluoro (has fluorine), D stands for deoxy (takes in oxygen), and G stands for glucose. The name of the chemical in this way indicates its structure and the name also gives a hint that FDG resembles glucose.
So what is 18F anyway? This 18F apparently holds the secret behind how it works as a “marker of light.” Actually, this 18F is what is called an “isotope” of the fluorine and this isotope is heavily involved with the phenomenon of gamma ray emission. Now let’s dive a little deeper into this topic, though it will be a little more specialized.
18F is a fluorine isotope

As we briefly touched on it above, the F in 18F is the symbol for the element fluorine. Here, let’s think about the structure of the fluorine atom.
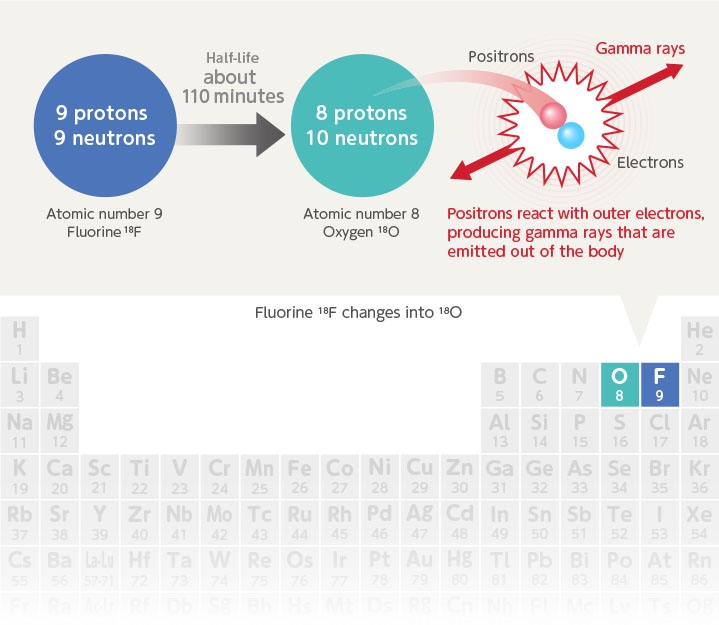
All atoms are made up of an atomic nucleus and electrons. The atomic nucleus is further made up of protons and neutrons. In other words, the atom is comprised of protons, neutrons and electrons. Among these, the “number of protons” determines the properties of the element. This is the quite familiar "atomic number" in the periodic table that indicates the type of atom. The atomic number 9 for fluorine shows that there are 9 protons (also same number of electrons).
However, there are atoms whose elements are the same yet have a different number of neutrons in the nucleus. These are called isotopes. Notation for the isotope usually shows the total number of protons and neutrons at the upper left of the element number and this is called the mass number. In other words, 18F shows a fluorine isotope with a mass number of 18.
All atoms are made up of an atomic nucleus and electrons. The atomic nucleus is further made up of protons and neutrons. In other words, the atom is comprised of protons, neutrons and electrons. Among these, the “number of protons” determines the properties of the element. This is the quite familiar "atomic number" in the periodic table that indicates the type of atom. The atomic number 9 for fluorine shows that there are 9 protons (also same number of electrons).
However, there are atoms whose elements are the same yet have a different number of neutrons in the nucleus. These are called isotopes. Notation for the isotope usually shows the total number of protons and neutrons at the upper left of the element number and this is called the mass number. In other words, 18F shows a fluorine isotope with a mass number of 18.
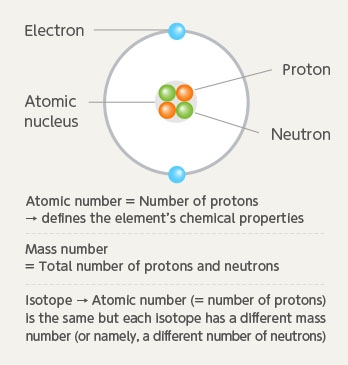
Atomic structure
18F is an unstable isotope

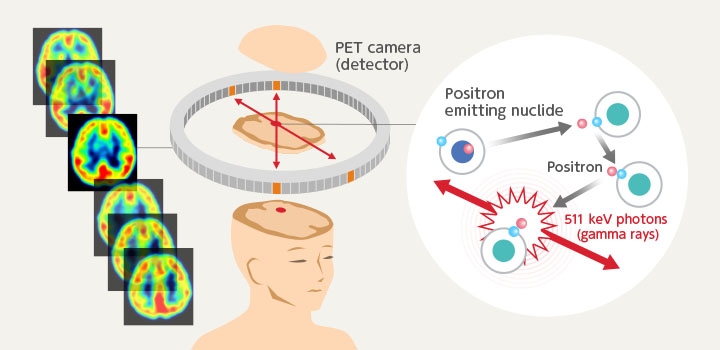
Some isotopes immediately change into another element and are called unstable isotopes. The 18F is also one of these unstable isotopes called radioactive isotopes. The isotope 18F emits positive electrons (called positrons which are particles having a positive charge and the same properties as an electron) and changes into 18O which is a stable isotope of oxygen. The positrons emitted at this time then bind to the surrounding electrons and emit light called gamma rays. In PET diagnosis these gamma rays are measured to search out cancer cells.
The “PET” in PET diagnosis is capital letters expressing “positron” for P, “emission” for E, and “tomography” for T.
The drawing below shows the reaction where 18F changes into 18O. A proton inside the 18F emits a positron and changes into a neutron. The 18F in this way loses 1 proton and the atomic number (or number of protons) decreases by one. In other words, the fluorine changes into oxygen that is one position to the left on the periodic table. The protons decrease but the neutrons increase, so there is no change in the mass number (total number of protons and neutrons).
The “PET” in PET diagnosis is capital letters expressing “positron” for P, “emission” for E, and “tomography” for T.
The drawing below shows the reaction where 18F changes into 18O. A proton inside the 18F emits a positron and changes into a neutron. The 18F in this way loses 1 proton and the atomic number (or number of protons) decreases by one. In other words, the fluorine changes into oxygen that is one position to the left on the periodic table. The protons decrease but the neutrons increase, so there is no change in the mass number (total number of protons and neutrons).
PET imaging

PET imaging
A unique feature of cancer cells is that they increase more rapidly than normal cells. This means they need large amounts of nourishment (glucose) and so take in more glucose and 18F-FDG that is an analogue of glucose.
Images captured by a PET camera show a lot of gamma rays (light) being emitted from cancer cells that have taken in large amounts of 18F-FDG. Cancer cells are detected by comparing these images with normal images and normal cells.
Images captured by a PET camera show a lot of gamma rays (light) being emitted from cancer cells that have taken in large amounts of 18F-FDG. Cancer cells are detected by comparing these images with normal images and normal cells.
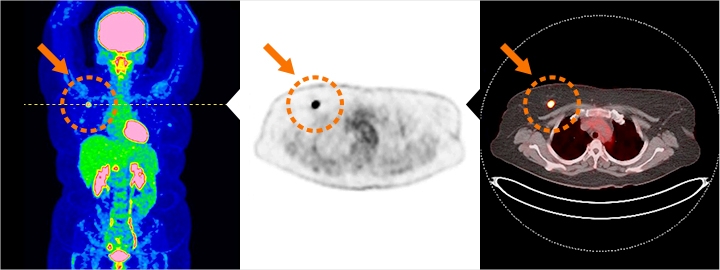
Left: Full-body PET image, Middle: PET image, Right: PET-CT fusion image
These are images of breast cancer (stage 1) detected early by PET screening. The small points shown by the orange arrows are FDG accumulations. By combining PET images with the images of the body structure such as CT images, even more information including the form, position and functioning (how the cells are functioning) of the cancer can be obtained to help detect cancer in its early stage.
18F produced nearby the PET Equipment

The time required for the constantly changing radioactive isotope to decrease to half of its former amount is called the half-life. The half-life of 18F-is about 110 minutes. Food stored in a refrigerator can last a long time, but the half-life of an isotope is almost totally unaffected by the outside environment. This means that the short-lived 18F-FDG for PET screening must be synthetically manufactured near the location where diagnosis takes place. Because of this, the 18F is produced by using a device called a cyclotron that accelerates particles within an electrical field to bombard protons onto special water containing 18O. Increasing the oxygen protons by 1 makes the number of protons reach 9 and the oxygen changes to fluorine of atomic number 9 (one neutron is also emitted at this time).

Cyclotron
On a final note…

Everything in our world is made up of a mere 118 elements. By discovering the wonderful principles of the periodic law, we have come to understand that the elements of the earth are finite and also how natural processes work.
As mentioned at the start, this year is International Year of the Periodic Table of Chemical Elements. Please take this opportunity to have fun and learn about the interesting elements, element symbols and the periodic table, and think about how we make use of these elements in our daily lives!
As mentioned at the start, this year is International Year of the Periodic Table of Chemical Elements. Please take this opportunity to have fun and learn about the interesting elements, element symbols and the periodic table, and think about how we make use of these elements in our daily lives!
Related information
International Year of the Periodic Table of Chemical Elements
https://www.iypt2019.org/
https://www.iypt2019.org/
See the other articles in this category.





